When a Bad Repair Leads to a Happy Ending: Homologous Recombination Deficiency Assays for Guiding Anti-Cancer Therapies
Genome instability - in which tumor cells sustain elevated frequencies of chromosomal lesions - is a fundamental property of cancer1. Such instability is often a driver of tumorigenesis and tumor progression. There can be many causes of genomic instability, but one of the key culprits is homologous recombination deficiency (HRD).
Homologous recombination (HR) is a highly conserved mechanism of repairing DNA double strand breaks. HR, even though it can be mutagenic2, is still good at repairing DNA faithfully, acting like an expert bicycle mechanic who fixes a broken chain without (usually) messing up anything else, like the gears. Take away or damage HR mechanisms though, and you get bad gears on your bike, er, that is, HRD. HRD-afflicted cells still need to repair DNA breaks or they’ll die, so cells turn to more error-prone mechanisms like non-homologous end joining (NHEJ) and microhomology mediated end joining (MMEJ)3. While many genes are required for homologous repair, the most well-studied are ATM, BARD1, BRCA1, BRCA2, CHEK2, BRIP1, MRE11A, NBN, PALB2, PTEN, RAD51C, and RAD51D.
So is HRD a bad thing? Yes, but it has an upside too, for patients with breast, ovarian and possibly also prostate cancer. High levels of genome instability are associated with improved outcomes for patients undergoing certain types of therapy. One such study showed that women carrying mutations in BRCA1 or BRCA2, genes that encode crucial components of the HR machinery, respond well to olaparib, a drug from the class known as poly (ADP-ribose) polymerase (PARP) inhibitors (PARPi), as well as platinum-based chemotherapeutics like cisplatin and carboplatin (reviewed in4). To capture more patients who can benefit from this treatment even if they do not carry mutations in the BRCA genes, additional predictive biomarkers were needed and HRD-induced genomic lesions met that need.
Next generation sequencing (NGS) is typically used to measure HRD. Copy number variation (CNV) microarrays are also employed since they are already widely used by cytogenetics labs and are relatively low cost, but the resolution of arrays is lower than NGS, is plagued by false positives, and is limited to known CNVs5.
To measure HRD, NGS data is put through an analysis pipeline that detects three genomic lesions, or “scars”6;
- Loss of heterozygosity (LOH), exemplified by loss of the wild type copy of a gene;
- Telomeric allelic imbalance (TAI) occurring in subtelomeric regions;
- Large-scale transitions (LST), which are CNVs of 10 or more megabases
Because HRD is a valuable predictor of response to PARPi and platinum-based therapies8, its measurements were implemented in many research and commercial assays. While Myriad Genetics’ NGS-based MyChoice HRD CDx designated assay has long dominated, many different assays, each with its own genomic instability metrics, followed (Table 1).
Most NGS assays measuring HRD are based on targeted NGS panels, usually combining mutational analysis of BRCA1/2 and other key HRR genes with SNP probes across the genome, which are used to detect the scars and calculate the instability metrics. However, other approaches using shallow whole genome sequencing (WGS) or a combination of both are also used, as well as applying custom analysis pipelines to CNV / SNP microarrays already widely utilized in cytogenetic labs. Genome instability measurements combine the 3 hallmarks of HRD in different ways or use a machine learning approach.
There is currently a strong push towards implementing HRD analysis as a distributed, local solution to reduce costs and wait times for patient treatment. However, it’s not a straightforward task to apply the results of different HRD analyses to treatment decisions. Although a consensus has been reached in the clinical community on the main hallmarks of HRD-related genomic scars, the different approaches to measuring them, calculating a genomic instability score, and determining the clinical cut-off, can introduce a high level of variability. The consensus is less clear on the clinical utility of measuring mutations in HRR pathway genes beyond BRCA1/2, but some assays also include them in their analyses.
Two major efforts to harmonize and align the rapidly growing field of HRD testing are currently underway (Figure 1). Since 2021, The Friends of Cancer Research (FoCR) have engaged with assay developers, patient advocacy groups and others to improve the use of HRD as a predictive biomarker by identifying best practices for HRD assay alignment and analytical validation. In Phase 1 of the project, FoCR surveyed the HRD assays on the market to better understand how HRD is defined and promote unified terminology9. Phase 2 compared the assay factors used to assess HRD and evaluated the level of concordance using a set of in silico ovarian cancer samples to identify the sources of variability10. Only moderate levels of concordance for measuring genomic scars were reported. Phase 3, aiming to put the findings in a clinical context, is expected to be finished by early 2024.
The second initiative comes from the German HRD Harmonization Consortium, a multi-center effort to compare both laboratory and analytical performance of decentralized HRD tests to the gold standard using a set of 100 patient FFPE samples. In addition, the study assessed inter-lab variability of each assay. The consortium has demonstrated substantial agreement in GIS scoring among most of the commercial and academic HRD assays tested, but only after applying statistical methods and extensive optimization11. Both reports highlight the need for further alignment and validation of HRD assays before their widespread clinical deployment.
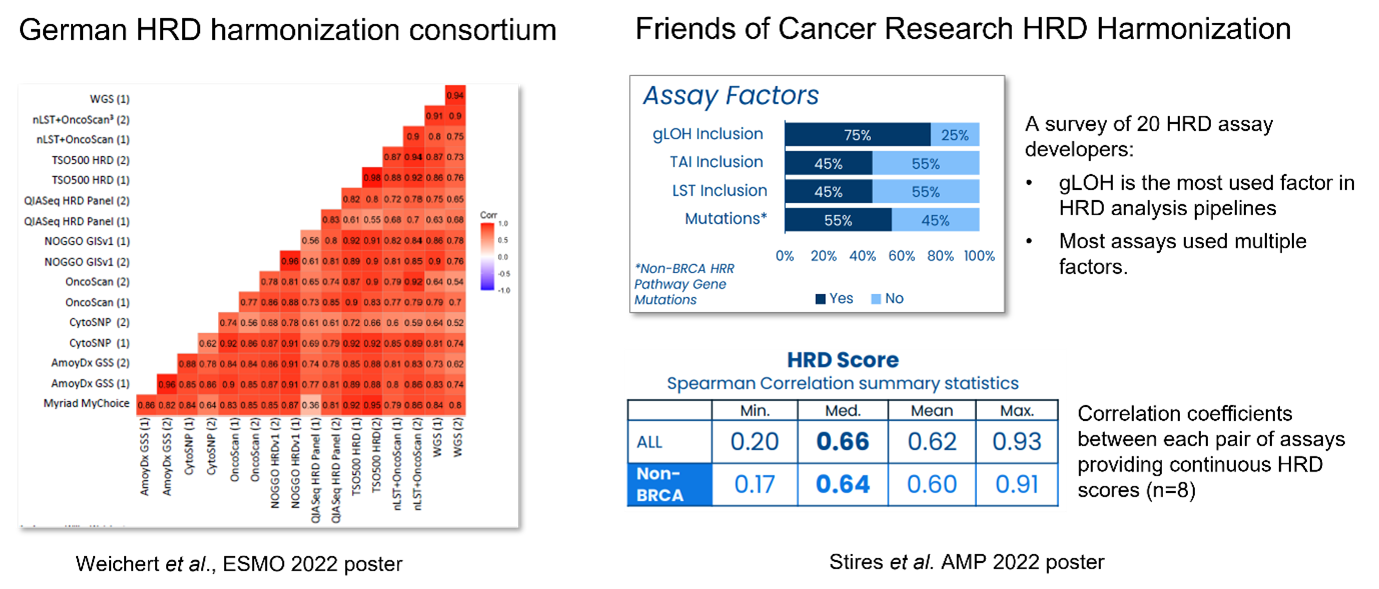
Figure 1. Major HRD harmonization efforts.
HRD is a complex biomarker, and NGS is a powerful tool to comprehensively characterize the resulting genomic scars. NGS-based assays require validation controls to gauge their reliability, and most assays on the market have been clinically validated using samples from the PAOLA-1 clinical trial with known Myriad GIS scores and clinical outcomes. However, well-characterized patient samples can be hard to access for labs who would like to establish and validate the performance of a distributed HRD solution in house.
Reliable determination of the copy number variation and SNPs across the genome, which is required to calculate the HRD genomic scar scores, becomes challenging in samples with low tumor content. It is therefore important to establish a limit of detection (LoD) of each HRD assay, which is difficult using clinical samples only.
LGC Clinical Diagnostics | SeraCare has created dedicated HRD reference materials to address these challenges. The Seraseq® HRD reference materials are derived from well-characterized cancer and SNP-matched normal cell lines. Two formats are available. One, a formalin-fixed, paraffin-embedded (FFPE) format, which controls for both DNA extraction from real-world patient samples as well as determining HRD scores, is made from cancer cell lines blended with matched normal cells at a tumor fraction of 65%. Purified genomic DNA from both the cancer and the normal line is also available as a matched pair that can be blended as needed to enable validation of new workflows and establishing the overall HRD score LoD. These materials also provide a high level of consistency. Because they have been made at very large scale, there will be little variation in GIS scores from lot-to-lot, which wouldn’t be the case if cells had to be re-grown from cryostocks when needed.
In both formats, three products with different GIS scores are available: Negative, Low Positive, and High Positive. The Negative and High Positive materials contain additional spiked-in plasmid-borne mutations in 4 HRR-related genes at variant allele frequencies (VAFs) useful for monitoring the quality of the sequencing data and relevant SNV LoD.
SeraCare’s HRD reference materials, the first commercially available reference materials dedicated to validation, implementation, and routine QC of HRD-focused assays, are an invaluable asset in the evaluation of cancer patients for life-saving treatments, and will play a critical role in the harmonization of HRD assays. Visit our website to learn more.
|
Platform |
Assay |
HRD metrics |
|
Targeted NGS |
BRCA1/2, GSS |
|
|
BRCA1/2 + LOH |
||
|
LOH + TAI + LST |
||
|
BRCA1/2, LOH + TAI + LST |
||
|
LOH + TAI + LST |
||
|
Not publicly available |
||
|
LOH + TAI + LST |
||
|
HRR genes + LOH |
||
|
HRR genes + LOH + TAI + LST |
||
|
LOH + TAI + LST |
||
|
Targeted NGS / Immunohistochemistry (IHC) |
30 HRR genes + LOH + TAI + LST (NGS) + PD-L1 (IHC) |
|
|
WES |
Not yet published |
|
|
Shallow WGS + targeted panel |
BRCA1/2 + CNV + LST |
|
|
BRCA1/2 + Machine learning |
||
|
OncoScan microarray |
Variable |
|
|
LST only |
||
|
LOH + TAI + LST |
Table 1. Commercial and academic HRD assays
NOTE: This blog was co-authored by Krystyna Nahlik, Ph.D.
References
- Hanahan, D. and Weinberg, R.A. (2011). Hallmarks of Cancer: The Next Generation. Cell 144, 646.
- Strathern, J. N., et al. (1995). DNA synthesis errors associated with double-strand-break repair. Genetics 140, 965.
- Bétermier, V., et al. (2014). Is Non-Homologous End-Joining Really an Inherently Error-Prone Process?. PLOS Genetics 10, e1004086
- Faraoni I. and Graziani G. (2018). Role of BRCA Mutations in Cancer Treatment with Poly(ADP-ribose) Polymerase (PARP) Inhibitors. Cancers 10:487
- Gabrielaite, M., et al. (2021). A Comparison of Tools for Copy-Number Variation Detection in Germline Whole Exome and Whole Genome Sequencing Data. Cancers 13, 6283.
- den Brok, WD., et al. (2017). Homologous Recombination Deficiency in Breast Cancer: A Clinical Review. JCO Precision Oncology 1, 13.
- Telli, M.L., et (2016). Homologous Recombination Deficiency (HRD) Score Predicts Response to Platinum-Containing Neoadjuvant Chemotherapy in Patients with Triple-Negative Breast Cancer. Clinical Cancer Research 22, 3764.
- Ray-Coquard, I., et al. (2019). Olaparib plus Bevacizumab as First-Line Maintenance in Ovarian Cancer. The New England Journal of Medicine 381, 2416
- Stewart, M.D., et al. (2022). Homologous Recombination Deficiency: Concepts, Definitions, and Assays. The Oncologist 27, 167.
- 2022 Research Partnerships Overview (friendsofcancerresearch.org)
- Weichert, W., et al. (2022). High concordance of different molecular assays in the determination of HRD associated GIS in high grade epithelial ovarian cancer. Annals of Oncology 33. DOI:https://doi.org/10.1016/j.annonc.2022.07.700



